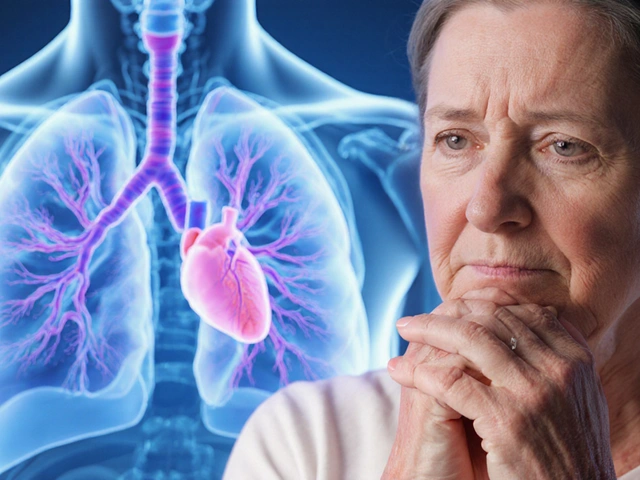
| Term | Focus | Example | Goal |
|---|---|---|---|
| Correction | Immediate Symptom | Wiping a spill off the floor | Stop the immediate issue |
| Corrective Action | Root Cause | Fixing the leaking pipe that caused the spill | Prevent recurrence |
| Preventive Action | Potential Risk | Replacing all old pipes before they leak | Prevent occurrence |
The CAPA Framework: The Gold Standard for Quality
In highly regulated fields, this process is formalized as CAPA is a system for Corrective and Preventive Action used to ensure regulatory compliance and product safety. Whether you are following ISO 13485 is the international quality management standard for medical device manufacturers or FDA guidelines, CAPA provides the roadmap to move from "something is wrong" to "this will never happen again." For those in the medical device or pharmaceutical worlds, CAPA isn't optional-it's a legal requirement. In fact, the FDA often issues warning letters to firms that treat CAPA as a paperwork exercise rather than a problem-solving tool. One major pitfall is addressing the symptom rather than the cause; statistics show that over 60% of firms fail to prove they've actually prevented a problem from returning during inspections.How to Execute a Corrective Action Process
Fixing a quality problem isn't about a lucky guess; it's about a disciplined workflow. Most successful manufacturers follow a six-phase approach to ensure nothing is missed.- Identification: Spot the event. This could be a customer complaint, a failed internal audit, or a sensor trigger on the line.
- Evaluation: Determine the risk. Is this a minor glitch or a critical safety failure? This categorization decides how much resource you pour into the fix.
- Root Cause Analysis: This is the hardest part. You need to dig deep to find why the error happened. Many use the 5 Whys is an iterative interrogative technique used to explore the cause-and-effect relationships underlying a particular problem or a Fishbone diagram to map out every possible variable.
- Resolution Planning: Create a plan. This must include specific action items, clear deadlines, and a named person responsible for the task.
- Implementation: Put the plan into action. This involves updating standard operating procedures (SOPs) and training staff on the new method.
- Effectiveness Review: This is where most fail. You must verify that the fix actually worked. This often requires statistical process control charts or testing a sample size of at least 30 units to ensure the result is statistically significant.

The Hard Truth About Root Cause Analysis
Root cause analysis is the engine of any quality system, but it's resource-heavy. On average, a significant non-conformity requires 8 to 12 hours of dedicated investigation. If your team is rushing through this in 30 minutes, they aren't finding the root cause-they are just guessing. For example, if a CNC machine is producing parts out of tolerance, a "quick fix" is to recalibrate the machine. But a root cause analysis might reveal that the floor vibrates whenever a nearby forklift passes by, causing the misalignment. Recalibrating the machine every hour is a correction; installing vibration-dampening mounts is a corrective action.Common Pitfalls and How to Avoid Them
Even experienced quality managers struggle with CAPA. The most common complaint is the mountain of paperwork. Some systems generate nearly 50 pages of documentation for a single issue, which can actually slow down the resolution process. To avoid these traps, consider these strategies:- Use Cross-Functional Teams: Don't let the quality department solve the problem in a vacuum. Bring in the operators who run the machines; they usually know why the error is happening before the engineers do.
- Digitize the Workflow: Moving away from paper logs to integrated digital tracking systems can reduce documentation time by about 41%.
- Tier Your Response: Not every glitch needs a full-blown CAPA. Create a tiered system where minor deviations get a simple correction, while critical safety issues trigger the full corrective action protocol.

The Future: AI and Predictive Quality
We are moving away from "detect and fix" toward "predict and prevent." The integration of Artificial Intelligence is technology that enables computers to simulate human intelligence to analyze patterns and predict failures is changing the game. Some manufacturers are already using AI to reduce investigation time by over 50% by spotting patterns in production data that humans would miss. By 2027, many experts predict that a majority of manufacturers will use predictive systems. Instead of reacting to a defect, the system will flag that a tool is wearing out or a temperature is drifting, triggering a corrective action before a single bad part is ever produced.What is the difference between a correction and a corrective action?
A correction is an immediate action to fix a detected problem (like replacing a broken part), whereas a corrective action identifies the root cause of that problem and implements a permanent change to ensure the problem does not happen again (like redesigning the part to prevent it from breaking).
How do I know if my corrective action was successful?
Success is measured through an effectiveness review. This involves monitoring the process over several production cycles to ensure the defect rate has dropped significantly-often by more than 50%-and using statistical validation to prove the change is permanent.
Why is CAPA so important for medical device companies?
Because medical devices directly impact patient safety. Regulatory bodies like the FDA require CAPA to ensure that any failure is thoroughly investigated and corrected. Inadequate CAPA systems are one of the top reasons for FDA warning letters.
What are the '5 Whys' in root cause analysis?
The 5 Whys is a technique where you ask "Why?" repeatedly (usually five times) until you reach the underlying cause of a problem. For example: The part broke. Why? Because it was brittle. Why? Because the heat treatment was too high. Why? Because the sensor was faulty. Why? Because it wasn't calibrated. Root cause: Lack of calibration schedule.
Can't I just use preventive action instead of corrective action?
Preventive action is for things that haven't happened yet. If you already have a quality problem, you need a corrective action to fix the existing failure. Preventive action is about analyzing risks to stop future failures before they ever occur.